|
NiMH
Battery State of Charge
Monitor
By: Dave Johnson |
|
Problem:
For the last 6 years or so, I have been using rechargeable NiMH
batteries for my MP3
players, my portable CD players, batteries for my digital cameras and AM/FM radios.
Rechargeable batteries make sense for devices which are used frequently. A good
battery can be recharged 100s of times. The ability to recharge a battery over and
over can really save a lot of money, not to mention less material sent to the dump.
But, lately I have been quite frustrated and angry at some of the rechargeable NiMH
cells I have purchased. |
 |
| The quality of these
devices seems to be getting worse and worse. The last round was a packet of 8
AA cells which I paid about $20 for. The batteries were made in China with
the American name “Duracell.” They were supposed to be rated at about 2.5
Amp-hours but although they were purchased new, they behaved like they were many
years old. With a good set of batteries installed, I can usually get to
play 6 or 7 CDs on my portable player before the pack of two AA cells would run
out of juice. The Duracell batteries died after playing only 2 CDs.
It was not just one bad cell in the bunch either. All the batteries had
the same low energy capacity. In contrast, I have some batteries which are
at least 5 years old and they are still going strong. How do you know when
you have a stinker in the bunch? |
| Typical Testing
Methods: The conventional way to measure the quality of a rechargeable
battery takes time. You first have to fully charge the device. Then,
you discharge it, usually at a constant current, and time how long the battery
can maintain that current before its voltage drops below a certain critical
cut-off point. In an AA battery, with a 2500 milliamp hour capacity, that
test could take a while, often days. In theory, with perfect battery, a 2.5
Amp-hour battery could support a 2.5 Amp load for an hour. But in
practice, the actual time would be much shorter. Internal resistance of
the battery prevents the efficient extraction of that energy. The 2.5
Amp-hour is usually defined with a much lower current, often down to 10
milliamps or so. At such a low current, it would take 250 hours or over 11
days complete a test. That kind of test would be impractical. It may
be possible to apply perhaps a 1 Amp load to the battery and compare how long
the battery supports that current to a known good battery. An hour long
test would certainly be better than one that takes over a week but it is still
way too long if you are trying to sort out some bad apples in a box of
batteries. I’d like to see if there might be a quicker way. |
| Proposed Faster
Testing Method: For many years I
thought that a pulsed dv/dI technique might be used to quickly determine the
health of a battery. This test involves drawing a heavy current pulse from
the battery and measuring the voltage change that results. This technique
in fact measures the internal resistance of the battery. A healthy
battery, when fully charged, should display a fairly low internal resistance.
As the battery ages, perhaps over the course of years of service, the internal
resistance should increase. But, the internal resistance will also
increase as the battery is discharged. So, this pulsed technique may be
used to weed out some fully charged but bad batteries and may also be used to
determine how much juice is left in the battery as it is discharged. The
first objective in this project is to come up with some statistics, so a typical
curve could be plotted showing the relationships between Amp-hour capacity, the
percentage of charge and the internal resistance. |
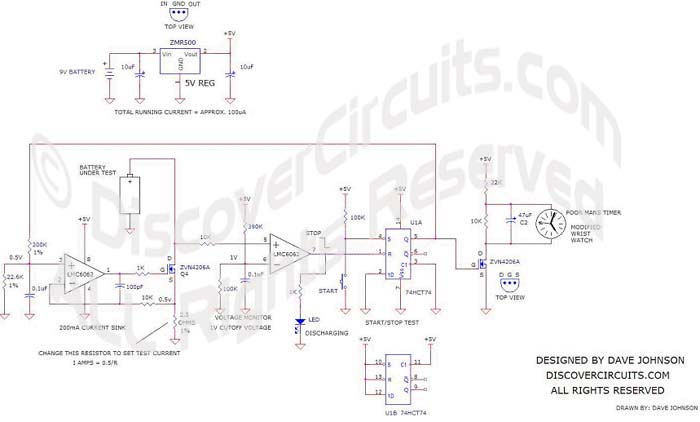
| Overnight Battery
Capacity Test: The circuit below
should work for testing some batteries over an 8 hour or so period. The
circuit draws a constant 200ma of current from a fully charged battery and times
how long the battery can support that current before its voltage drops below
1.0v. I’ll use a cheap clock or watch, set up as an elapsed timer. I
expect that a good cell will run for perhaps 8 to 10 hours. I should be
able to test about two cells per day with this circuit. |
|
|
|
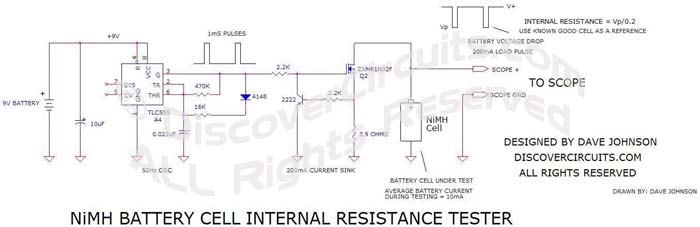
Delta V Battery
Resistance Test Circuit: The first objective for this round of
experiments is to see how consistent the internal resistance of some batteries
is, once fully charged. I think I will first try using the circuit shown
below. This simple circuit will pulse the battery with 1ms 200ma pulses. I
will use my scope to measure the voltage drop in battery voltage with each
pulse. I’ll first charge up a bunch of batteries then measure the
resistance with this circuit. I then will try to develop a relationship
between the measured internal resistance and the measure Amp-hour capacity of
the cell. I would expect to see a higher resistance for those cells which
have a lower Amp-hour capacity. After those tests, the last step in the
process would be to discharge batteries to some repeatable point, and try and
come up with a relationship between state of charge and internal resistance.
That might take a bit of head scratching. I will share with you my test
results. |
|
 |
|
|
|
|